THE HAMLIN GROUP
SPECIALIZING IN COMPUTATIONAL CHEMISTRY SINCE 2018
I strive to empower those around me with the confidence to advance the scientific field, each and everyday.
I motivate my students and colleagues to solve problems, which likely seem tough while they do it, but
after they make a breakthrough and unravel the problem, I think they feel invincible.
I motivate my students and colleagues to solve problems, which likely seem tough while they do it, but
after they make a breakthrough and unravel the problem, I think they feel invincible.
|
TREVOR A. HAMLIN
ASSISTANT PROFESSOR Department of Theoretical Chemistry Amsterdam Institute of Molecular and Life Sciences (AIMMS) Amsterdam Center for Multiscale Modeling (ACMM) Vrije Universiteit Amsterdam 1081 HV Amsterdam The Netherlands E-mail: [email protected] TREVOR'S ACADEMIC FAMILY TREE |
ANNOUNCEMENTS
NEW TheoCheM Website
Today, on 23 March 2021, we celebrate the launch of the new TheoCheM website. From now on, this is the official website for all information about our team working on Molecular Theoretical Chemistry.
Today, on 23 March 2021, we celebrate the launch of the new TheoCheM website. From now on, this is the official website for all information about our team working on Molecular Theoretical Chemistry.
CBOND2020 is postponed to September 2022. Because of the Corona crisis, the 3rd European Symposium on Chemical Bonding, CBOND2020, which was to take place originally from 22 till 25 September 2020 in Amsterdam, The Netherlands, has been postponed to September 2022. We aim at keeping both time schedule and venue unchanged and in the same week of September but, at this point, we have to renegotiate with our campus organization. More information is published as soon as the new reservation has been completed.
The primary objective of CBOND2020 is the exchange of the latest developments and ideas in the field of chemical bonding. On Monday 21. 09. 2020, the day before the main conference begins, CBOND2020 will have a school for PhD students on Kohn-Sham molecular orbital (MO) theory and the associated canonical energy decomposition analysis (EDA), as implemented in the ADF program. This school will have lectures in the morning and a hands-on computer lab in the afternoon.
RECENT NEWS
Paradigm-shifting discovery by myself, Israel Fernandez, and Matthias Bickelhaupt on dihalogen catalysis of Michael addition reactions, published in Angewandte Chemie, covered by Chemistry World in article by Katrina Krämer: "Quantum mechanical mechanism behind 100-year-old textbook reaction revealed".
Today, the 7th of May, 2019, Diego Cesario has successfully defended his thesis “Insights from a Theoretical Approach on the Weak Intermolecular Forces: the Transition from Van Der Waals to Chemical Bond”.
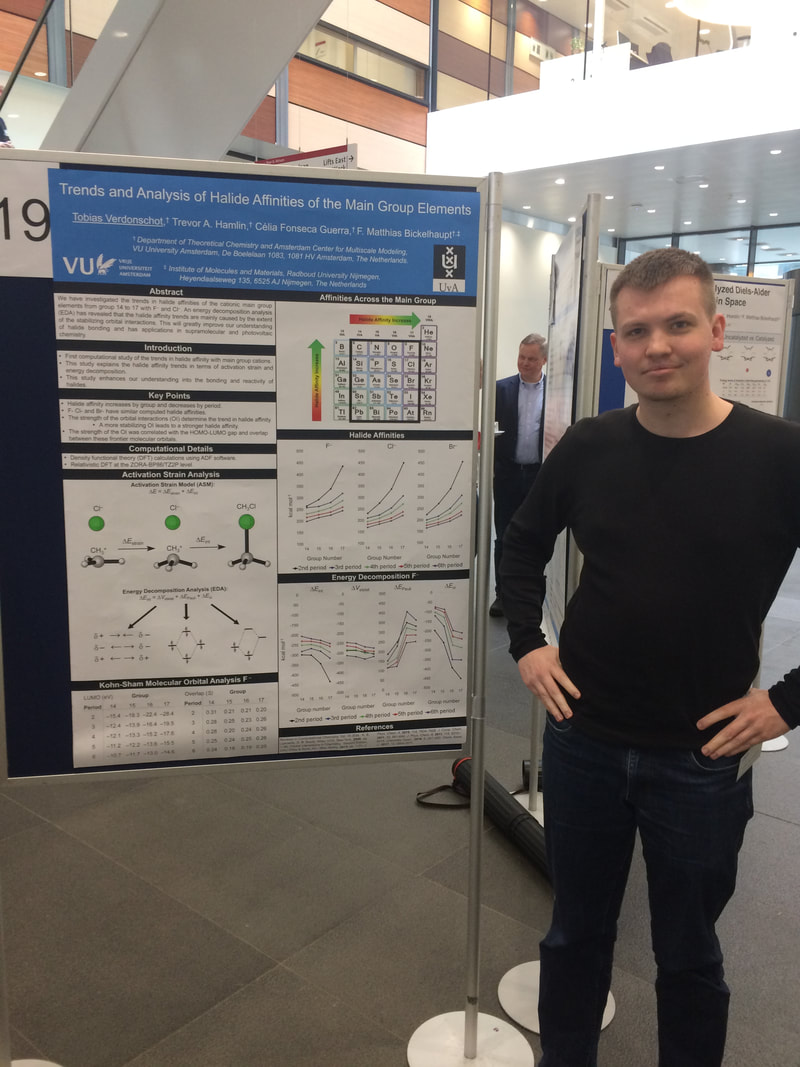
On 26 March 2019, the first annual symposium of the Computational and Theoretical Chemistry (CTC) Division of the Koninklijke Nederlandse Chemische Vereniging took place in the Auditorium of the O|2 Building at Vrije Universiteit Amsterdam. With around 120 participants, top speakers, and 50+ excellent posters, it has been a great day!! I delivered an invited lecture and we had a number of outstanding poster presentations from the group members! Here are some photos of the day.
HBOND2019
|
The 23rd International Conference on "Horizons in Hydrogen Bond Research" (HBOND2019) will be held from September 24 till September 27 in Amsterdam, The Netherlands.
We have an outstanding program with speakers from all around the world. I hope to see you in Amsterdam! WEBSITE |
Second European Symposium on Chemical Bonding in Oviedo, Spain!
Thanks for Célia Fonseca Guerra for the photograph!
Highlight on VU Website and Newsletter
|
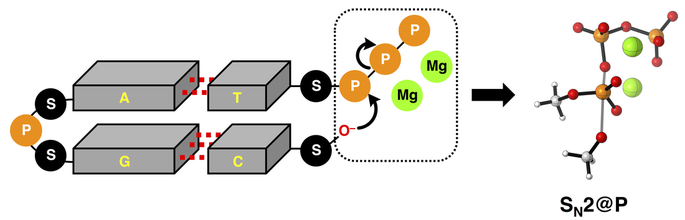
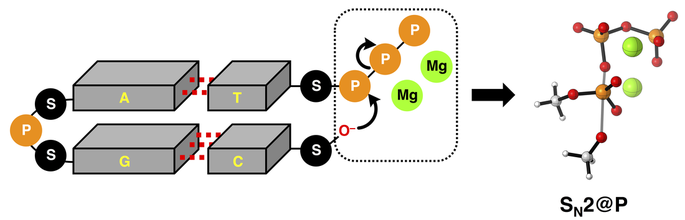
From the news article: "VU Theoretical chemists Trevor A Hamlin and Matthias Bickelhaupt have revealed how magnesium ions lower the reaction barrier in enzymatic triphosphate hydrolysis (a key step in DNA replication and other reactions that are vital to life), using quantum chemical methods." Read the full article HERE.
|
Full paper in Chemistry – A European Journal!
|
Barrier or no barrier? A full account on the solvent-induced changes to the potential energy surface (PES) of a variety of nucleophilic substitution (SN2) reactions is reported. We provide simple guidelines, based on differential solvation of the electronic structure of a reaction system, to understand when and how minima and maxima appear along the PES.
|
Review Article on SN2 Reactions in ChemPhysChem!
|
The reaction potential energy surface (PES) and, thus, the mechanism of bimolecular nucleophilic substitution (SN2) depend profoundly on the nature of the nucleophile and leaving group, but also on the central, electrophilic atom, its substituents, as well as on the medium in which the reaction takes place. In this review article, we provide an overview of recent studies and demonstrate how changes in any one of the aforementioned factors affect the SN2 mechanism.
|
Paper in Chemical Communications!
|
I could not be more proud of this paper, as it mark my first one as a corresponding author! It is a key work in the study of SN2@P reactions, which are key in DNA replication or transcription concominant with backbone elongation in the newly forming DNA or RNA strand. We explore the effects of triphosphate deprotonation and elucidates the physical factors underpinning the enzymatic role of Mg2+ ions on this pivotal SN2@P reaction.
|
Paper in JOC in collaboration with the Houk Group at UCLA!
|
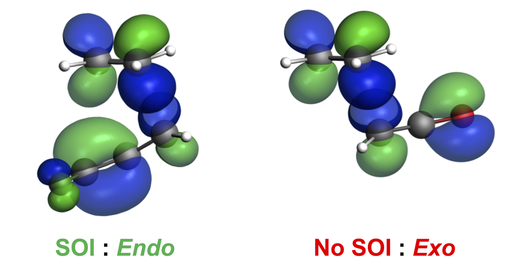
The endo and exo stereoselectivities of Diels–Alder reactions of cyclopropenone, iminocyclopropene, and substituted triafulvenes with butadiene were rationalized using density functional theory calculations. We highlight the role of secondary orbital interactions and charge transfer in determining the stereoselectivity of this transformation!
|
Paper in JOC with the Rutjes Group at Radboud University!
|
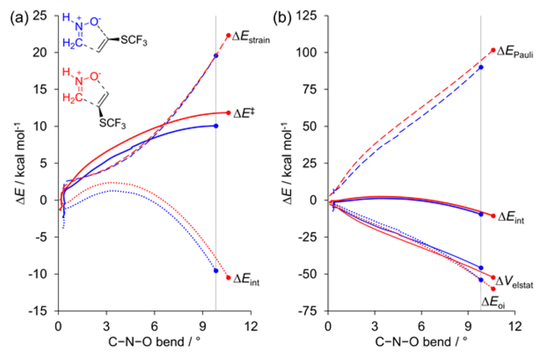
First and foremost, a well deserved shout out to Bas van Beek on his first (of many) paper in my group! I just love collaborating with experimentalists and this paper in JOC was just that. Description without jargon: Collegues from the group of Floris Rutjes at Radboud University and Bayer CropScience have discovered a way to synthesize molecules that have a really cool group on them (-SCF3). These compounds have potential applications as drugs and agrochemicals. My group modeled the reaction on a supercomputer and determined the reason why they obtained only 1 of 4 possible products: steric interactions drive regioselectivity. This is a big step towards a more rational design of novel chemicals in the future!
|